Schlagwörter
Aktuelle Nachrichten
America
Aus Aller Welt
Breaking News
Canada
DE
Deutsch
Deutschsprechenden
Global News
Internationale Nachrichten aus aller Welt
Japan
Japan News
Kanada
Karte
Karten
Konflikt
Korea
Krieg in der Ukraine
Latest news
Map
Maps
Nachrichten
News
News Japan
Polen
Russischer Überfall auf die Ukraine seit 2022
Science
South Korea
Ukraine
UkraineWarVideoReport
Ukraine War Video Report
United Kingdom
United States
United States of America
US
USA
USA Politics
Vereinigte Königreich Großbritannien und Nordirland
Vereinigtes Königreich
Welt
Welt-Nachrichten
Weltnachrichten
Wissenschaft
World
World News
2 Kommentare
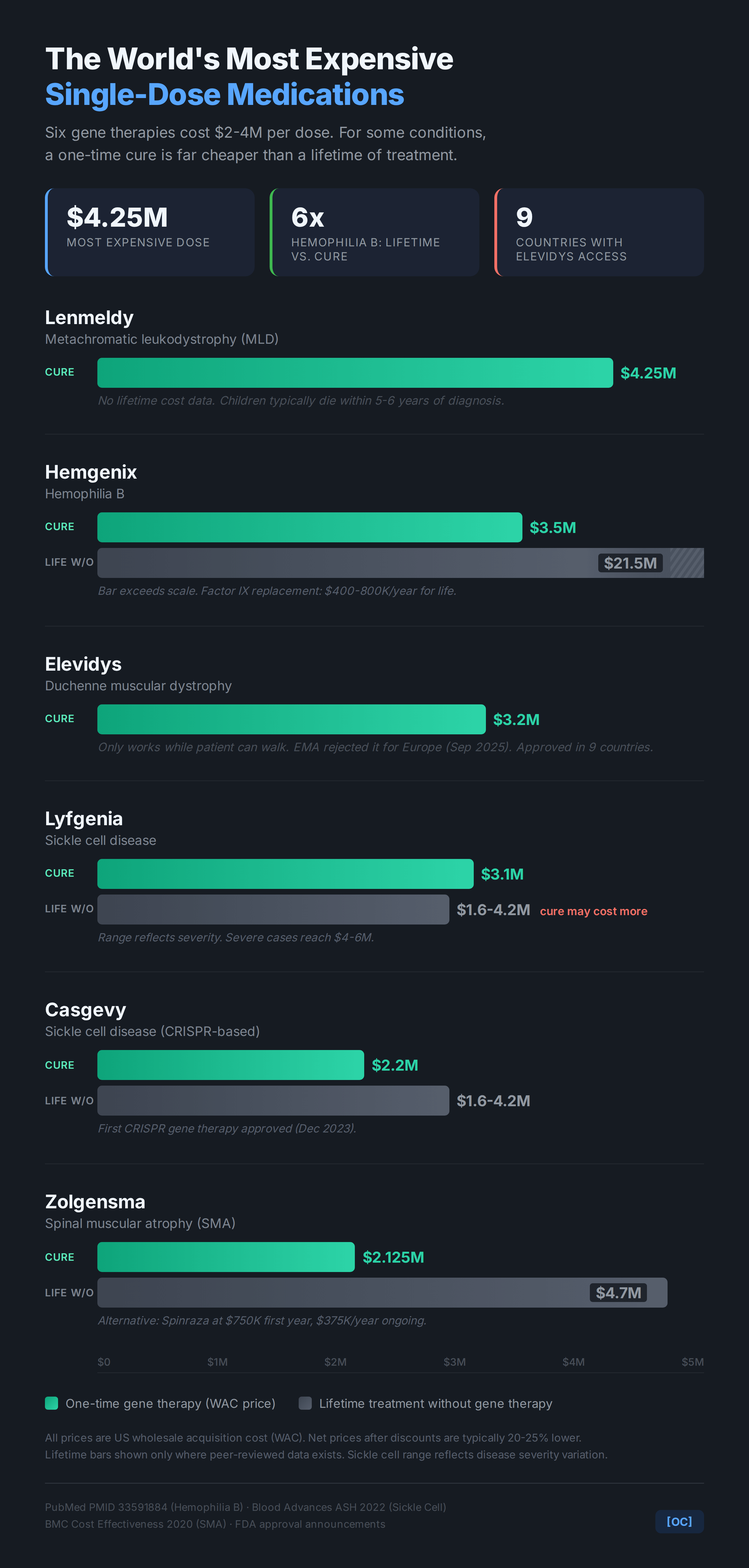
Prices are US wholesale acquisition cost (WAC) from FDA approval announcements and manufacturer press releases. Net prices after rebates are typically 20-25% lower. Lifetime costs from peer-reviewed literature:
* Hemophilia B lifetime: $20-23M, midpoint $21.5M used in chart (PubMed PMID 33591884, adult payer perspective)
* SMA/Spinraza: $750K first year, $375K/yr ongoing. $4.7M figure reflects ~11 years of treatment (BMC Cost Effectiveness and Resource Allocation, 2020)
* Sickle cell: $1.6-4.2M depending on severity (Blood Advances, ASH 2022)
Lifetime comparison bars only shown where solid peer-reviewed data exists. For Lenmeldy (MLD) and Elevidys (Duchenne), I couldn’t find reliable lifetime cost estimates in the literature, so those only show the gene therapy price.
Tool: HTML/CSS, screenshotted at 3x
I’ve read that there’s still a bit of an issue with the uptake and prescription of a lot of these drugs. It seems the economic argument is undeniable and yet they’re not being widely rolled out? I wonder if there’s lobbying and public relations campaigns from pharmaceutical companies that produce prophylaxis treatments that are wildly profitable? I get the district feeling that the last thing a lot of pharmaceutical companies want is to cure diseases. Why have a $4 million one off when you can have a $25 million lifetime customer?