Schlagwörter
Aktuelle Nachrichten
America
Aus Aller Welt
Breaking News
Canada
DE
Deutsch
Deutschsprechenden
Global News
Internationale Nachrichten aus aller Welt
Japan
Japan News
Kanada
Karte
Karten
Konflikt
Korea
Krieg in der Ukraine
Latest news
Map
Maps
Nachrichten
News
News Japan
Polen
Russischer Überfall auf die Ukraine seit 2022
Science
South Korea
Ukraine
Ukraine War Video Report
UkraineWarVideoReport
United Kingdom
United States
United States of America
US
USA
USA Politics
Vereinigte Königreich Großbritannien und Nordirland
Vereinigtes Königreich
Welt
Welt-Nachrichten
Weltnachrichten
Wissenschaft
World
World News
3 Kommentare
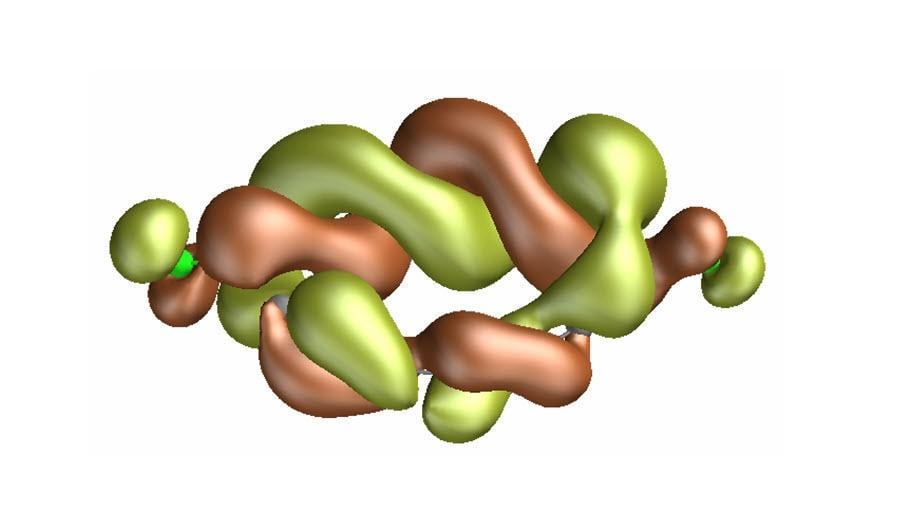
> A recent paper in the journal Science announced the discovery of something genuinely and strangely new that could have a huge impact on our ability to chemically engineer advanced materials. A team of quantum scientists from IBM, the University of Manchester, Oxford University, ETH Zurich, EPFL and the University of Regensburg created and characterized a new molecule unlike any other — with a quirk in its shape that can be turned on and off to change how electrons corkscrew through it and alter its chemical behavior.
>This experiment wasn’t the result of trying to incrementally improve an existing molecule. It created a brand new form of matter never before synthesized, observed or predicted. The new molecule’s chemical formula sounds innocent enough: C13CL2. That means it is composed of 13 carbon atoms and two chlorine atoms. That is an unremarkable-sounding formula for such an unusual chemical compound. But what C13CL2 does with its electrons is not only stunning, but unlike anything we have seen before. And it begins with its exotic topology.
>This breakthrough has the potential to impact several important domains. Molecules with a topological state that can be flipped on demand could be the basis of entirely new classes of switches, sensors or information storage media. Even more tantalizing is the potential impact on drug discovery. Exploring molecular properties with quantum computing has long been touted for that purpose, but the quantum computing simulation pipeline tested on C13CL2 could represent a future workflow in which new drug candidates can be modeled at the electronic level with a fidelity far beyond that of classical computers. If that is possible, it could eliminate years of trial-and-error currently required for pharmaceutical development.
So if I understand correctly, they actually did perform computational chemistry on a quantum computer here. it’s not just a toy example or a demonstration of some “impossible” molecule. They’re extracting meaningful electronic structure information, not just showcasing a contrived system.
Article is written in a misleading way but anyhow, it’s impressive.
I am really confused with the title and the summary, did they use a quantum computer to discover the molecule?